

A supercritical fluid (SCF) is any substance above its critical temperature and pressure; the fluid has the properties of both gas and liquid while not exhibiting a distinct liquid or gaseous phase. Thus, an SCF is,
• Dense like a liquid to dissolve materials
• Has low viscosity, high diffusivity, no surface tension like a gas
Supercritical Fluids:
The Technology
Carbon dioxide is the most commonly used supercritical fluid and becomes supercritical at 31oC and 74 BAR. There are many applications for scCO2 outside the textile dyeing industry, most commonly used in decaffeination of coffee and extraction of natural herbs and spices.
Carbon dioxide is environmentally friendly, non-toxic and in our case most of the CO2 is internally recycled with very little vented to the atmosphere. scCO2 based textile operations represent a very “green” technology.
For more information about the systems, visit the
Applied Separations website.
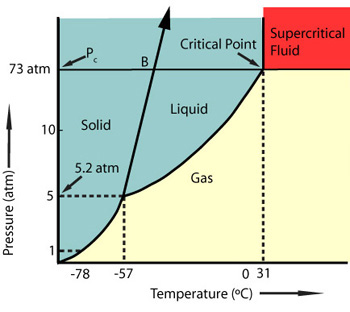
Phase Diagram for Carbon Dioxide